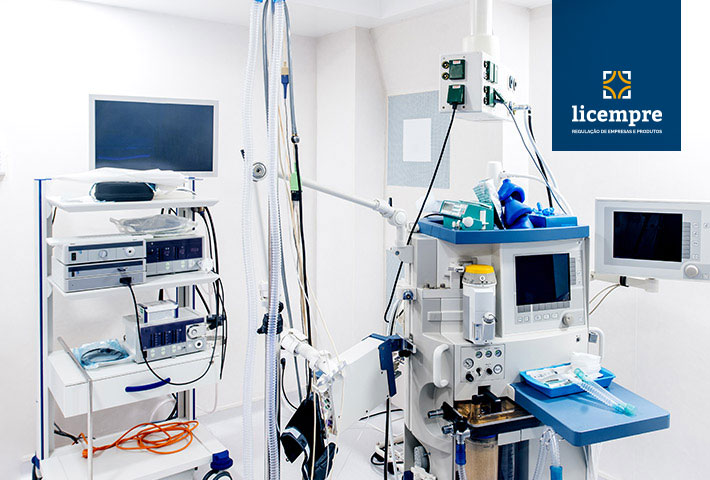
The regularization of healthcare products such as hospital equipment is done at ANVISA.
Thus, for the product to be exposed for sale or consumption it is necessary to register with the Ministry of Health, following the requirements and determinations provided for in the legislation.
Regularization of hospital equipment at ANVISA

Hospital equipment under the Sanitary Surveillance regime includes all equipment for medical, dental, laboratory or physical therapy purposes used directly or indirectly in the diagnosis, therapy or rehabilitation of persons.
Therefore, no health-related product, national or imported, can be industrialized or marketed in the Brazilian market without registration with the Ministry of Health. Only in view of the exception of those mentioned in § 1 of Article 25 of the aforementioned Law, that although dismissed are still subject to the Sanitary Surveillance regime.
As a result, failure to comply with the foreseen determinations generates infringement with the Federal Sanitary Legislation being the infringing company, subject to the penalties provided for in Law No. 6.437, of August 20, 1977.
Above all, before requesting the regularization of hospital equipment at ANVISA, it is necessary to evaluate the steps presented below in order to verify all documents and information to be inserted in the process. Check out the 5 steps for the regularization of hospital equipment at ANVISA:
Step 1 – Regularization of the Company with Health Surveillance
The first step for product registration or registration of this hospital equipment at ANVISA comprises obtaining the Operation Authorization – AFE, from ANVISA and the Operating License. Thus, without proper authorization the protocol of the petition is not possible.
- Local Operating License – LOL
The LOL is issued by the local Sanitary Surveillance, whether municipal or state according to the company’s location.
- Good Manufacturing and Control Practices – BPFC
Good Manufacturing and Control Practices are comprised of requirements set forth in RDC Resolution No. 16 of March 28, 2013. Therefore, where every company that intends to manufacture, import or market medical devices must follow these requirements.
Step 2 – Sanitary Identification of Equipment
The Sanitary Identification of Medical Equipment is related to its identification and classification in ANVISA criteria. Thus, the equipment is analyzed for certification and reporting needs to obtain registration.
- Equipment Classification
Equipment classification is a step where it allows the process to be conducted in the correct manner, as documentation varies according to equipment type and classification.
- Economic Information Report
Some medical equipment is subject to presentation of the Economic Information Report pursuant to ANVISA Resolution No. 3385 of October 13, 2006.
- INMETRO Certificate of Conformity
The INMETRO Certificate of Conformity is also required on some medical devices following the criteria stated in Normative Instruction No. 04 of September 24, 2015.
Step 3 – Petition Identification
When identifying the petition it is necessary to evaluate some points based on the previous sanitary identification. Therefore, some of these points in question is to evaluate if the product is subject to registration or registration with ANVISA.
If you already have a record, which petition is desired as (change, cancellation, revalidation, transfer of ownership, amendment or rectification of publication).
- Register or registration?
The regularization of medical products at ANVISA allows two types of regularization: register and registration.
Registration is a simplified procedure and is applicable to Class I and II products belonging to the small and medium risk classes.
Medical equipment subject to registration falls into risk classes III and IV, high and maximum risk classes.
- Single Equipment or Equipment Family
The process may refer to a single piece of equipment or a family of pieces of equipment.
The equipment family includes several models in the same process. Thus, all models in the family must belong to the same manufacturer.
Step 4 – Electronic Petitioning
Electronic Petitioning is an action that effectively initiates the request for registration or registration of a product in ANVISA. In cases of complementary petition it should also be performed.
Thus, the process of electronic petition is done under a registration at ANVISA performed by the portal itself with the requested information. Therefore, in order to perform the electronic petition, the company must have performed the classification and the type of petition identification that it wishes to perform.
Step 5 – Petition Protocol
Finally, the Petition Protocol is performed after completing the process assembly and performing the electronic petition. The petition must be physically filed at ANVISA.
The Petition, when filed must be signed by its legal and technical requesting company applicants, as determined by art. 6 of Law No. 9,784, of January 29, 1999.
Licempre is a company specializing in regulatory services and serving the entire product registration process with ANVISA.


